Material for energy conversion, storage or efficiency
Energy and environmental challenges imply a more efficient and sustainable managment of energy conversion and storage. Within this framework, the OXYDES team characterizes new structures or architectures -when talking about ceramics and thin films- of materias able to convert (themoelectric conversion per example), or store energy.
Finding new structures with adequate properties will allow the team to build among others : cathods for fuel cells, micro-betteries, micro-supercapacities 2D and 3D.
Tools and methods
Materials for energy conversion
Thermoelectric materials are able to transform directly heat into electrical energy or inversely electrical energy in cold (Seebeck and Peltier effects) without any emissions (CO2, other gases, radiations, …), vibrations or moving parts. Oxides have attracted interest of the thermoelectric community, in 1997, when Terasaki et al. (Physical Review B 56 (1997) R12685) have reported a large Seebeck coefficient in the metallic oxide NaxCoO2. Moreover, in a recent paper based on abundance as well as economy Herfindahl−Hirschman indices for production and reserve, Gaultois et al. (Chem. Mater. 25 (2013) 2911) have concluded that only highly electrically conductive transition metal oxides, silicides with low bismuth content, and Half-Heuslers alloys combine good thermoelectric performance and availability.
The team uses two strategies to deal with all the aspects of the thermoelectric oxides research :
Improvement of thermoelectric properties of known oxides ceramics :
The studies are focused on Ca3Co4O9 for p-type materials and ZnO and SrTiO3 for n-type materials.

Well known thermoelectric ZnO nanostructured by SPS at 600°C.
Research of new thermoelectric oxides :
Many oxide systems and new concepts are explored ( oxygen deficient (K0.5Na0.5)NbO3 , Pseudobrookite (Fe2TiO3), Ba2Co9O14 ).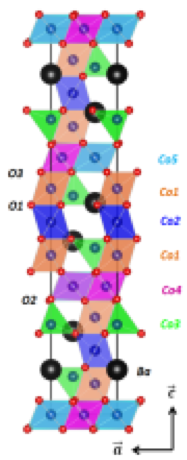
Ba2Co9O14 cristallographic structure, studied as new thermoelectric material.
SOC cells are electrochemical devices that convert the chemical energy of a fuel into electricity. With regard to electrodes, the development of materials with mixed conduction properties is a key issue for improving SOCs performance at high temperatures. To increase their efficiency, a particular attention must be paid on the development of oxygen electrode materials. Lanthanum strontium cobalt ferrite, LaxSr1-xCoyFe1-yO3-δ (LSCF) is one of the most developed oxygen electrode used in the high temperature (800°C - 1000°C) solid oxide cells. Various compositions were tested on cell deposition by screen printing.
Fuel cell structure. A schematic of fuel cell in which the layers (anode (Ni-YsZ), electrolyte (8-YsZ), cathode (LSCF) and ceramic separator (CGO) are deposited on a cermet support.
Materials for energy storage
The over the last few years the development of miniaturized power sources and the diversification of devices, has increased the need of three-dimensional (3D) integration. As an example, components such as Metal/Insulator/Metal (MIM) and Micro-Electro-Mechanical-Systems have been developed with 3D integration to be embedded into chips. Theses 3D- developed components have also been widely developed for applications such as Dynamic Random Access Memory [1-3] optical or Radio-frequency (RF) switches The benefits of 3D architectures are the reductions in size, cost, and the increase in reliability and energy related performance.
Moreover 3D structuring enhances the physical properties of devices. Therefore research in this area is extremely active, particularly in the fields of micro-batteries, supercapacitors, fuel cells , solar cells. This is about 20 to 30 times superior to the two-dimensional (2D) thin films capacity. In the area of RF applications, the integration of functions requires more complex grouping of active and passive components.
Current deposition techniques for growing thin films on 3D substrates are divided in two categories: physical and chemical. Physical techniques consist in Physical Vapor Deposition (PVD), vacuum evaporation, molecular beam epitaxy and sputtering. These techniques require combining work (high voltage, vacuum, etc.) and low deposition rates (close to 10 nm.min-1). Chemical techniques consist in electro-deposition by electrophoresis, Chemical Vapor Deposition, the plasma activated chemical vapor deposition, Plasma-Enhanced Chemical Vapor Deposition, atomic layer depostion (ALD),….We demonstrated in our laboratory a new approach to the production of three-dimensional-coated patterns using liquid route. Metallic perovskite oxides were coated onto three-dimensional (3D) microstructured substrates with different aspect ratios. The success of the method relies on the solution viscosity monitored by adding viscous liquid. The process of oxide thin films consists in three steps: preparing the precursor solution, coating the solution by spin-coating process onto three-dimensional-Si substrates and post-annealing. The chemical solution 3D-coating is conformal.

Cross-sectionnal SEM micrograph of a TiO2 coating in plot Si, with aspect ratio 25.
The TiO2 layers have been deposited on silicon pillars for microsupercapacitor electrode.
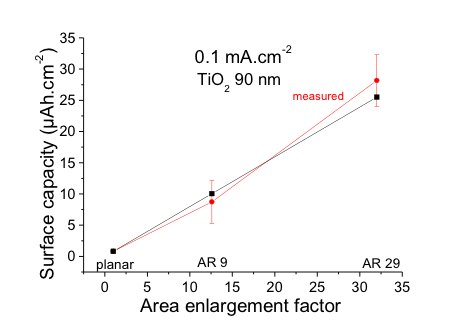
Capacities measured are plotted as a function of the surface area enlargement factor. The capacities measured are plotted as a function of the surface area enlargement factor. The capacity is correlated with the area enlargement factor and measured capacity corresponds to expected value, which means that the entire active material layer takes part in the electrochemical process.
For microbatteries applications, silicon nanoparticles SiNPs are coated by liquid route onto 3D silicon substrate for negative electrode. These SiNPs layers onto 3D structure have an impact on the electrochemical performances and its preparation and deposition improvement is a key component to maximize performances in Li-ion microbatteries.

SEM images of trench structures for aspect ratio of 3 after SiNPs deposition.
The electrochemical properties are correlated with the area enlargement factor.

